FreeWeigh.NET for Statistical Quality Control
Comprehensive - Flexible - Modular - Scalable - Networking
Universal, most powerful Statistical Quality Control (SQC) for any industry
FreeWeigh.NET for Statistical Quality and Process Control is a PC based software solution that allows to control and fine tune perfectly customer production process. Modular license model allows system adoption to any needs. Flexibility and ease of use allows further optimization of production, increase quality and productivity.
With a Web-base Test Place Module customer will be able to run a test module and move across the production plant with one mobile PC, using a standard web browsers and without additional sofware installation.
Furthermore it is the ideal solution to collect any attributes of quality data and enables use of Testplans. Watch the video and imagine what other quality measures could you controll using FreeWeigh.Net?

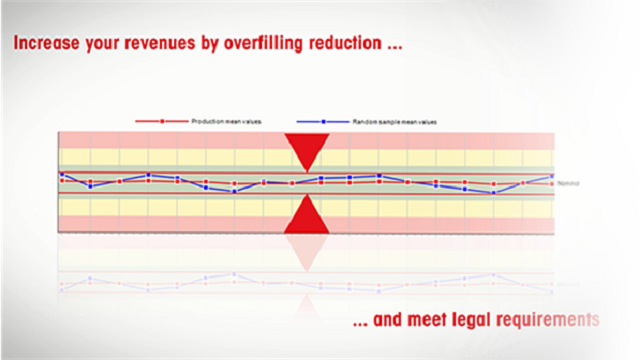
Compliance and production cost control
FreeWeigh.Net provides all relevant tools to avoid underfilling and comply with legal legislations, including its Audit Trail and 21 CFR Part 11 Modules. At the same time production process is still monitored to reduce undesired overfilling and provide your production cost optimization.
Check out the product in action and listen customer experience
This short video shows a FreeWeigh.Net solution working in the real environment. Get a first impression of the product capabilities and learn more about benefits it can bring to your production.

Comprehensive overview of quality processes
Use Statistical Process Control (SPC) to get early information about process deviations. Ensure targeted process results to be kept as specified.
Validation of Weighing Processes
Freeweigh.Net analyzes weighing data to continuously monitor critical process parameters. Comply with specific regulations regarding validation of computerized systems and handling of electronic records and signatures.














