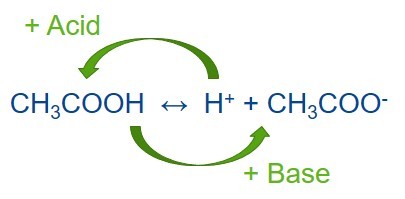
Buffer preparation is a common process in chemistry and biochemistry laboratories. A buffer solution is a mixture of a weak acid and its conjugate base or a weak base and its conjugate acid. Buffer solutions are used to help maintain a stable pH value of another solution that is mixed with the buffer. The buffer resists changes in the pH value of the whole solution when a small amount of a different acid or alkali is introduced into the solution either through addition or via any chemical reaction within the solution. Buffer solutions are therefore very useful in a wide variety of applications in which a relatively stable pH is required. A buffer may also be called a pH buffer, hydrogen ion buffer, or buffer solution.
For example, blood contains natural buffers to maintain a stable pH of between 7.35 and 7.45 so that our enzymes work correctly. As enzyme activity varies with pH, maintaining a constant pH is essential in biochemical assays to ensure the correct level of activity is observed. In commercial applications, buffers can be found in shampoos to prevent skin irritation, in baby lotions to inhibit the growth of bacteria, and in contact lens solutions to ensure the pH level of the fluid remains compatible with that of the eye.
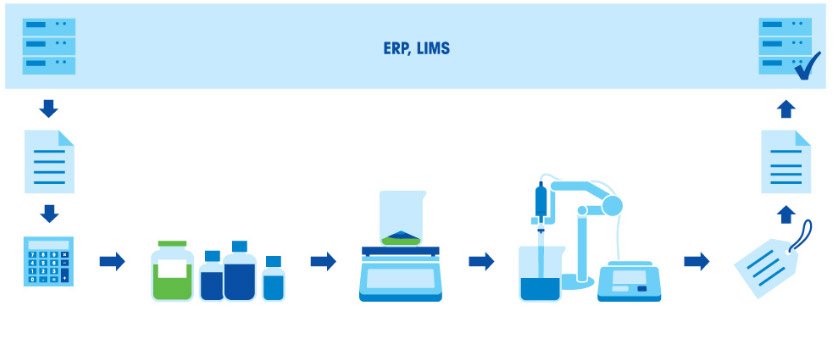
Preparing buffers consists of several steps: Weighing-in the components, dissolving the components, adjusting the pH and replenishing to the final volume. As the ratio of the acid to base in a buffer is directly related to the final pH, it is vital to weigh the components with a high degree of accuracy. So, it is important that the equipment used (balance, pipettes and pH meter) are properly calibrated and have sufficient accuracy.