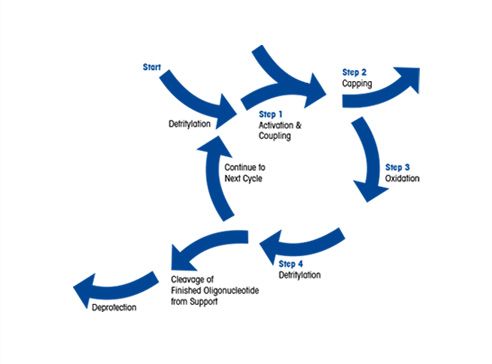
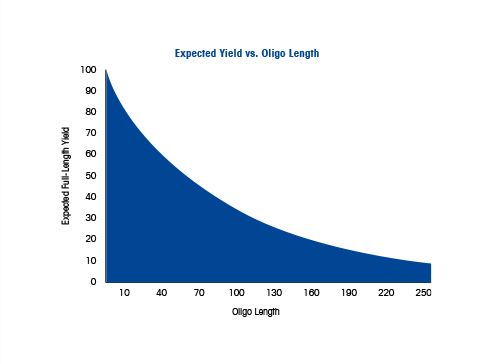
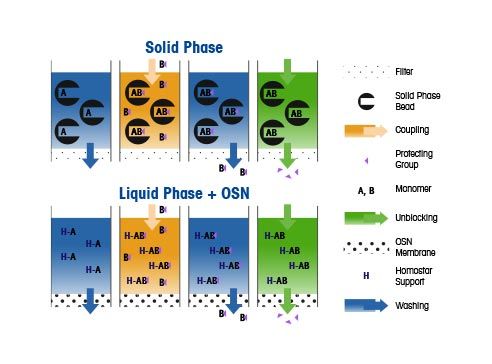
Oligonucleotide Primers and Probes
The most common use of synthetic oligonucleotides is as relatively short probes and primers (up to 30-mer) in a wide variety of applications. This involves synthesizing a nucleotide sequence that is paired or 'reverse-complimentary' to a larger, target DNA or RNA strand (target sequence). As primers, oligos are typically used to initiate enzymatic reactions to e.g. create millions to billions of copies of a short or long target sequence. Well-known examples are the polymerase chain reaction (PCR) or the Sanger sequencing method. Applications for oligo primers include DNA sequencing, gene expression, cloning and molecular diagnostics.
As probes, oligos serve to identify and bind to a specific DNA or RNA target sequence in order to confirm presence of this sequence in a given material. Applications using oligo probes include blotting procedures such as northern blotting (for RNA) or southern blotting (for DNA), as fluorophore-conjugated sequences in microarrays that detect changes in genes expression or used in screening for genetic diseases or to identify specific pathogens (molecular diagnostics).
Oligo Therapeutics/Gene Therapy
In therapeutic applications, antisense oligonucleotides (ASO), generally 20 to 30-mer species, take advantage of natural biology and facilitate gene inhibition or gene silencing (destruction) of undesirable or over-active RNA sequences, this in turn blocks expression of certain damaged or overactive proteins which may be causing or facilitating disease. Research on oligonucleotide based therapeutics has intensified tremendously and several drugs have been approved in recent years.
Future Use of Synthetic Nucleotides: Exploring DNA and RNA Vaccine Modalities
Although not an oligonucleotide by strict definition, DNA- or RNA-based vaccine products, such as mRNA or plasmid or vector-based nucleic acids, of many hundreds or thousands of bases in length, represent the frontlines of evolving synthetic nucleotide technologies.
In concept, DNA or RNA vaccines would dispense with all unnecessary or harmful parts of a pathogenic bacterium or virus. Instead, such a nucleic acid-based vaccine would contain code for just a few parts of the pathogen's DNA or RNA. These DNA or RNA strands instruct the patient's own body to produce individual antigens or fragments of the pathogen, and then promote an immune response to the antigen. With modern computing and in silico modeling, these oligonucleotide vaccine modalities are capable of being created in a matter of days or weeks given an appropriate target sequence to design against. As a platform technology, nucleic acid-based vaccines rely on standard sets of building blocks or raw materials enabling myriad of combinations almost at will. As such, they are also relatively cheap and easy to produce compared to traditional vaccine modalities. However, this is still a maturing paradigm for the biopharmaceutical industry and new challenges are constantly being addressed, some unique to oligo and long nucleic acid products and some shared with other biotherapeutic modalities.