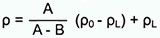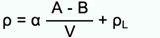Measuring density of a sample is an important quality parameter of both raw materials and finished products. Various techniques enable the density of solid, viscous and liquid materials to be accurately determined e.g. metals, plastics, chemicals, lubricants and food.
Density for Quality Control
A variation in a raw material, indicated by a change in the density, may have a detrimental outcome on the functioning or quality of the final product. Density measurement of raw materials can be used to confirm the purity of the material. If a substance has been adulterated with a cheaper alternative, the measured density of the composite material will be different from the pure substance.
Density can also be used to ensure homogeneity. If a manufactured part is not homogenous, key performance attributes such as strength and crack resistance can be affected. For example, an internal air bubble could ultimately cause a part to fail when placed under stress. Random sampling of parts is a simple and cost-effective way to monitor ongoing quality.
Why Accurate Weighing is Crucial
Common laboratory gravimetric procedures for determining density are the buoyancy technique, the displacement principle and the pycnometer method.
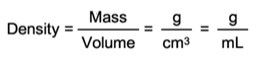
The most widely used method is the buoyancy technique which utilizes Archimedes' principle: a body immersed in fluid indicates an apparent loss in weight equal to the weight of the fluid it displaces. This ancient principle, from around 200 BC, is precisely what is used nowadays to determine density gravimetrically. Accurate density measurement therefore is highly dependent upon accurate weight values.

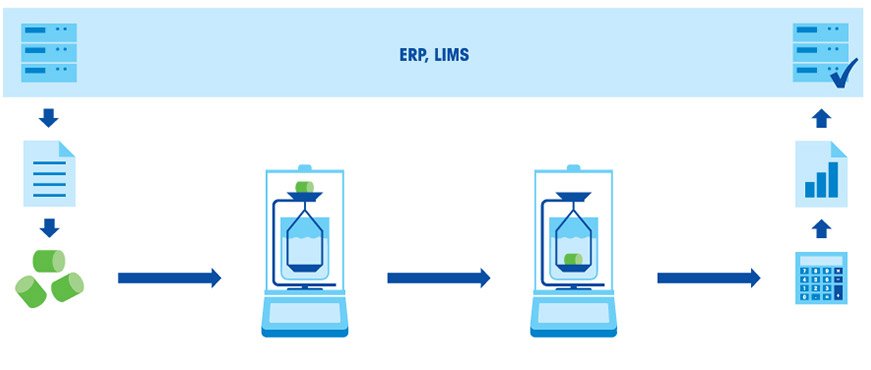
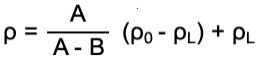
 Beaker for auxiliary liquid stands on a platform or below the balance.
Beaker for auxiliary liquid stands on a platform or below the balance. Beaker for auxiliary liquid stands on the balance.
Beaker for auxiliary liquid stands on the balance. Glass beaker of defined volume.
Glass beaker of defined volume.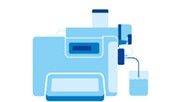 Oscillating tube technology
Oscillating tube technology