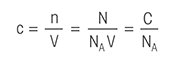The following titration definitions may make it easier for you to learn all you need to know about titration.
Analyte: Specific chemical species in a sample of which the content or amount is determined by titration.
End of titration: The desired endpoint or equivalence point at which titration is terminated and titrant consumption evaluated. More than one equivalence point may occur during the same titration.
Endpoint: The point at which the end of the titration reaction is observed (typically by color change or another titration indicator). To define titration along with its endpoint represents the classical technique.
Equivalence point: The point at which the number of entities (equivalents) of the added titrant is the same as the number of entities of sample analyte.
Indication: The procedure to follow the reaction and detect the end of the titration. Generally, indication is made using potentiometry (electrodes) or color indicators.
Parallax: The difference in the apparent position of an object based on the observer’s line of sight. In titration, this phenomenon must be considered when visually observing liquid menisci in a burette.
Primary standard: A certified, highly pure substance used for the accurate determination of titrant concentration.
Signal acquisition: Signal acquisition is the monitoring of physical phenomena to obtain digital or numeric values at certain points (such as a titration experiment endpoint or equivalence point).
Standardization: The use of a highly pure reference chemical substance (standard) to determine titrant concentration.
Stoichiometry: The mole/mass relationships between reagents and products in titration. Titration reagents react according to fixed relationships, so if the amount of separate reactants is known, the amount of the product can be calculated. If the amount of one reactant is known and the amount of products can be determined, the amount of other reactants can also be calculated.
Titrant: A solution of a certain chemical reagent that is standardized in terms of concentration so it can be used for accurate titration.
Titration: A quantitative chemical analysis in which a defined amount of titrant reacts quantitatively with the sample compound being analyzed. From the volume of titrant consumed, the amount of sample compound is calculated based on the stoichiometry of the assay reaction. Also known as volumetry or titrimetry.
Titration curve: Titration curves illustrate the qualitative progress of a titration. The curve generally uses the volume of the titrant as the independent variable and the solution as the dependent variable. A curve allows rapid assessment of the titration method. Curves take four forms: symmetric, asymmetric, minimum/maximum, and segmented.
Titration cycle: The cycle that is performed and repeated until the endpoint or the equivalence point of the titration reaction is reached. The titration cycle consists primarily of four steps: titrant addition, titration reaction, signal acquisition, and evaluation.
Titration equation: A series of titration formulas that allow the calculation of the mass molarity of solid samples, the concentration of acid and base solutions, the concentration of diluted solutions, and the titrant consumption after a direct titration. For easy calculation of titration results, visit our Titration Calculator.
Titration theory: Titration theory explores the determination of a product related to its concentration by observing a titration reaction using either physical or electrochemical methods. Knowing the exact concentration of a species, product, or chemical function helps to ensure process efficiency and/or product quality.