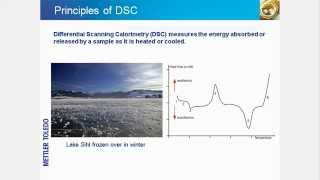
In this tutorial, you will learn about the principles of DSC and its sensor technology, measurement possibilities, plus DSC industries and applications. These include isothermal curing, temperature modulated DSC, oxidation induction time (OIT), crystallinity, glass transition temperature, oxidation onset temperature (OOT), degree of cure and kinetics, safety studies and polymorphism.
Visit us for more information on DSC (Differential Scanning Calorimetry) and other thermal analysis solutions, as well as promotions, webinars, trainings, applications and handbooks
In addition to DSC, we also offer tutorials on these thermal analysis techniques: