 |
Table of Contents:
TA Tip
- Interpreting DSC curves; Part 2: Isothermal measurements
New in our sales program
- Crucible brochure
- Automatic liquid nitrogen refilling system
Applications
- Characterization of petroleum products with DSC
- Applications of DSC to thermosetting materials
- Force and temperature modulated TMA measurements of fibers
- Measurements of pore size distribution with DSC
- Measurements of low concentrations of PE-LD in PE-HD
- OIT of polyethylene with the TMA/SDTA840
Tips and hints
- Effect of sample mass on TG results
Characterization of petroleum products with DSC
Introduction
Conventional fuel is obtained exclusively from petroleum or crude oil. Petroleum is primarily a mixture of 6 different classes of substances. The composition of the mixture is specific to the region where the oil occurs and consists of
- straight-chain n-alkanes (CnH2n+2 ) with molar masses between 16 and 300 g/mol
- branched-chain alkanes (iso-alkanes)
- cycloalkanes
- aromatics
- sulfur-containing compounds
- polycyclic and heterocyclic resins as well as bitumens with molar masses of about 1000 g/mol.
Distillation of the crude oil yields various fractions, which are classified as follows: low boiling fractions, e.g. gasoline (petrol), aviation gasoline, naphtha; higher boiling fractions, e.g. fuel or heating oil and diesel; and high boiling fractions (heavy oil and lubricating oils). The residue after distillation is known as bitumen (or asphalt).
In the liquid state, the distillate appears macroscopically as a single-phase mixture. On cooling, crystals are formed, i.e. a multiphase mixture is obtained. The separation of crystalline material is undesirable and leads to a number of problems:
- Crystallized material separates out forming a sediment. This is often a problem, especially for the storage of diesel and heating oils.
- Crystallized material is retained in fil- ters, which can lead to blockages.
- Bitumen (asphalt) products are mainly used for surfacing roads. Crystallization causes the surface to become brittle and results in the formation of cracks.
Hydrocarbon distillates consist primarily of complex hydrocarbon compounds and crystallizable fractions. The former are partially liquid at room temperature and exhibit a glass transition at low temperatures. The glass transition temperatures of the liquid constituents depend on the petroleum distillate. Typical values are -30 ° C for bitumen, -130 ° C for diesel and -150 ° C for gasoline. The proportion of the crystallizable fractions is between 0% and 10% for bitumen, between 5% and 25% for fuel oil and up to 40% for crude oil. The chemical structure of the crystals depends on the distillate. With fuel oils, n-alkanes with 10 to 28 carbon atoms crystallize out, with bitumen n-alkanes with 20 to 60 carbon atoms; and with crude oil n-alkanes with 5 to 60 carbon atoms. Lightly branched iso-alkanes and cycloalkanes are also present.
[…]
Applications of Differential Scanning Calorimetry to thermosetting materials
Introduction
Differential Scanning Calorimetry (DSC) allows endothermic or exothermic enthalpy changes in a sample to be quantitatively measured. Melting processes in partially crystalline plastics, glass transitions and chemical reactions such as the curing of thermosetting resins can all be routinely analyzed [1].
The heat capacity increases on heating above the glass transition. The glass transition temperature, Tg , and the change of the specific heat capacity at the transition, ∆cp , are characteristic of the state of a plastic. If intramolecular and intermolecular mobility become restricted, for example as a result of increased crosslinking, then the glass transition temperature increases. ∆cp allows certain conclusions to be drawn about molecular interactions.
Prepregs are semi-finished products made from fabric impregnated with resin hardener mixtures. They are used to manufacture thermosetting molded materials through the action of pressure and temperature [2]. During the course of the development of a resin system for SMC prepregs (Sheet Molding Compounds) on the basis of unsaturated polyester resins (UP resins), the following questions arose concerning:
- the control of reactivity, e.g. whether the desired degree of cure is achieved even if the formulation is varied,
- the optimum duration of the compression (i.e. molding) and curing operations, and
- how long the prepregs can be stored, i.e. their storage stability.
[…]
Literature
[1] B. Benzler: Dynamische Differenzkalorimetrie - Hohe Reproduzierbarkeit Plastverarbeiter 47 (1996) 9, Seite 66
[2] B. Benzler: Vollständig vernetzt? Dynamische Differenzkalorimetrie an EP-Harzen Plastverarbeiter 47 (1996) 11, Seite 58
Force and temperature modulated TMA measurements of fibers
Intruduction
Thermal shrinkage is an important property of fibers that depends on the structure of the individual fibers concerned and on the processing conditions used, i.e. thermal history [1]. Shrinkage can be measured by classical Thermomechanical Analysis (TMA) using a low tensile force (load) at constant heating rate. The linear coefficient of expansion αe of a sample that does not shrink can be determined in the same way. Young’s modulus (E) can also be measured with TMA by modulating the tensile force during the heating program [2].
With drawn (stretched) fibers, however, the expansion that occurs on heating is superimposed on the shrinkage and cannot therefore be measured directly by TMA. Expansion and shrinkage are processes that are physically linked via the structure of the sample. When structural relaxation occurs, both properties change, but at different rates, because they are controlled by different effects (e.g. free volume and crystal perfection). In addition, shrinkage is influenced by irreversible changes in the internal stress. As a result of this, αe changes on shrinking, and the original thermal expansion coefficient of a stretched fiber cannot therefore be determined in a second heating measurement.
A special technique, known as temperature modulated TMA, is used to simultaneously determine expansion and shrinkage behavior. In much the same way as temperature modulated DSC (ADSC), a modulated heating rate is superimposed on the mean heating rate. As a result of this, the temperature oscillates sinusoidally, e.g. it oscillates up and down every three minutes with a maximum of 5 K/min and a minimum of -4 K/min at a mean rate of 1 K/min. The sample shrinks slowly and irreversibly due to the increase of the mean temperature. The normal reversible expansion, however, follows the small temperature changes. This means that αe can be determined from the oscillating changes in length induced by the temperature modulation independent of the shrinkage coefficient αs [3].
Young’s modulus (E) is determined from the change in length that occurs when the tensile force is modulated. If the frequency of this modulation is appreciably higher than that of the temperature modulation, then both excitations can be applied simul- taneously. Fourier analysis is then used to calculate E, αe and αs from the TMA measurement curve.
[…]
Literature
[1] M. Jaffe, Thermal Characterization of Polymeric Materials, 2nd Edition, E.A. Turi Ed., Academic Press, New York 1997, 1767.
[2] H. G. Wiedemann, R. Riesen, A. Boller, ASTM STP 1136, American Society for Testing and Materials, Philadelphia (1991) 84.
[3] R. Riesen, J. E. K. Schawe, J Thermal Anal. 59 (2000) 337-350.
Measurement of pore size distribution with DSC
Introduction
Pore size distribution (PSD) is a critical property of many materials. Ceramics, catalysts, pharmaceuticals, and the author’s own special area of interest namely cellulosic pulp fibers, are examples of such materials. The classical methods for measuring pore size distribution are gas sorption and mercury porosimetry. An interesting alternative technique is based on DSC and is commonly known as thermoporosimetry. In this article, thermoporosimetry refers to the measurement of pore size density based on the determination of the melting point depression of an absorbate held in a porous material.
[…]
Measurement of low concentrations of PE-LD in PE-HD
Introduction
Small amounts of PE-LD are often added to PE-HD to modify its mechanical properties. The following experiments describe the use of DSC to determine the detection limit for such additions, and to investigate the extent to which these additions can be quantitatively determined in polymer mixtures. Four samples of different (known) PE-LD content were used for these experiments. The samples of pure PE were first measured. Figure 1 shows that the melting behavior of PE-LD differs clearly from that of PE-HD. Assuming at least a partial incom- patibility of the two materials, one can expect the DSC melting curve of mixture of PE-LD and PE-HD to exhibit two clear melting peaks, as shown in Figure 1.
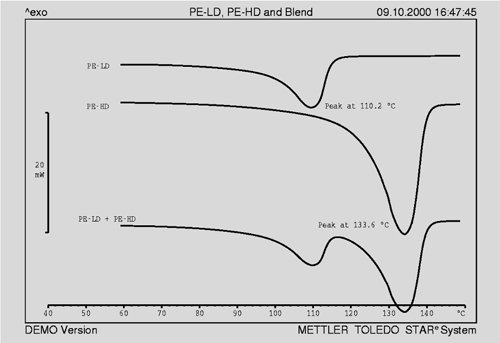 |
Fig. 1. Melting curves of PE-LD and PE-HD. The peak maximum of the PE-LD melting curve is appreciably lower. The crystallinity of PE-LD and PE-HD is also different; typically the degree of crystallinity of PE-HD is about 65%, and PE-LD about 25%. |
[…]
OIT of polyethylene with the TMA/SDTA840
| Sample | Crosslinked PE from a plastic pipe (PE-X) | |
| Information expected | OIT, Oxidative Induction Time at 210°C in oxygen | |
| Measuring conditions | Measuring cell | TMA/SDTA840 with Gas Controller at the reactive gas inlet |
| Probe | Negative load of -0.01 N, raised, (no changes in length can be measured with the probe raised) | |
| Sample preparation | A piece of ca.15 mg was cut off with a knife | |
| Crucible | Light aluminum pan, 20μl, with no lid | |
| TMA measurement | Heating from 50° C to 210°C at 20 K/min, then isothermal for 5 min under nitrogen. Afterward, switched to oxygen (gas inlet: reactive gas) |
[…]




